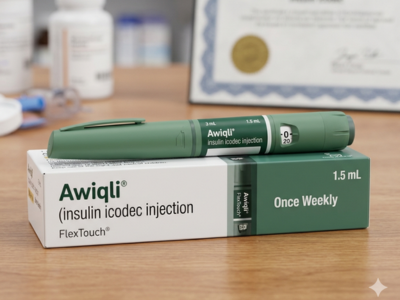

The U.S. Food and Drug Administration (FDA) has approved Awiqli, representing a major milestone in diabetes care by enabling once‑weekly basal insulin therapy for adults living with type 2 diabetes. This new treatment, developed by Novo Nordisk, marks the first and only long‑acting basal insulin product designed for weekly use in adult patients, and it is poised to transform routine glucose management.
Novo Nordisk revealed that the FDA green‑lit Awiqli (insulin icodec‑abae) injection, 700 units/mL, as an adjunct to diet and exercise for improving glycemic control in adults with type 2 diabetes. The regulatory decision underscores the FDA’s recognition of the need for flexible, patient‑centric insulin regimens that can better align with diverse lifestyles.
Awiqli’s approval has emerged after rigorous evaluation, including the ONWARDS phase 3a clinical program, in which the once‑weekly treatment demonstrated efficacy comparable to that of daily basal insulin therapies. Across multiple randomized and active‑controlled trials, Awiqli showed meaningful reductions in glycated hemoglobin (HbA1c) when used either with mealtime insulin or in combination with common oral diabetes agents and GLP‑1 receptor agonists.
Healthcare professionals have welcomed the approval of Awiqli as a milestone that addresses a long‑standing need for less frequent dosing options. Patients with type 2 diabetes often struggle with the burden of daily injections, and a weekly regimen could significantly reduce treatment complexity and enhance adherence over time.
Anna Windle, PhD, Group Vice President of Clinical Development, Medical & Regulatory Affairs at Novo Nordisk Inc., emphasized that Awiqli offers a meaningful alternative to traditional daily basal insulin therapies. She noted that simplifying treatment from seven daily injections to a single weekly dose could improve the overall care experience for adults with type 2 diabetes.
According to company statements, the safety profile of Awiqli through the clinical program was consistent with the basal insulin class. Commonly observed adverse events included episodes of hypoglycemia (lowered blood sugar), injection‑site reactions, allergic responses, skin thickening or indentations at the injection site, itching, rash, and potential weight gain.
Experts believe that the introduction of Awiqli may encourage broader conversations about insulin adherence, particularly among patients who have delayed treatment initiation or struggled with daily therapy regimens. Because Awiqli is administered once weekly, it could benefit individuals who find frequent injections burdensome or disruptive to daily life.
Novo Nordisk has a longstanding reputation in diabetes care, with a robust portfolio that includes established insulin products and novel therapies. Awiqli’s approval reinforces the company’s commitment to innovation and patient‑focused solutions in metabolic disease management.
Looking ahead, Novo Nordisk plans to launch Awiqli in the U.S. market, following final preparations and commercialization strategies. Once available, the product is anticipated to provide clinicians and patients with practical alternatives for optimizing long‑term glycemic control.
Regulatory setbacks in the past had delayed earlier progress for once‑weekly basal insulin formulations. Notably, previous attempts to secure FDA approval faced hurdles, including complete response letters and requests for additional information related to manufacturing and indications. These earlier challenges were addressed through comprehensive data submissions and trial refinements, ultimately paving the way for success with Awiqli.
More than 40 million Americans have diabetes, approximately 1 in 8, with 90% to 95% affected by type 2 diabetes. While type 2 diabetes typically develops in adults over 45, rising cases are now reported in children, teens, and young adults, highlighting an urgent need for flexible treatment options like Awiqli.
The U.S. Centers for Disease Control and Prevention estimates that millions of adults in the United States live with type 2 diabetes, and many struggle to maintain optimal glucose control due to treatment complexity and lifestyle factors. With Awiqli, healthcare providers now have a powerful tool to tailor treatment plans to patient preferences, potentially enhancing outcomes and quality of life.
In addition to its clinical benefits, Awiqli’s once‑weekly regimen may also reduce the overall treatment burden on healthcare systems by decreasing the frequency of prescription refills and associated medical visits. This shift could prove particularly meaningful in settings where frequent clinic attendance is challenging for patients.
The approval of Awiqli highlights the evolving landscape of diabetes therapy, where innovations increasingly emphasize convenience, precision, and patient engagement. As more data emerge from real‑world use and long‑term follow‑up, clinicians and patients alike will gain further insights into how weekly basal insulin fits within broader care strategies.
Overall, the FDA’s authorization of Awiqli represents a significant advance in diabetes treatment, offering adults with type 2 diabetes a new option that combines clinical efficacy with the convenience of once‑weekly dosing. The diabetes community is poised to evaluate the tangible impact of this innovation, with hopes that it may enhance treatment satisfaction and improve long‑term outcomes for many patients.